India Cardiac Surgery Site is associated with experienced cardiologists to deliver the perfect treatment and recuperative plan. Before the surgery, we will educate the patient with every fact involved in surgery and maintain the transparency in procedure, facilities and the related costs. We provide quality services and also assists with arrangements by keeping the concerns of the international patients in mind, providing you the utmost care and professionalism.
How to Get Started?

Planning your medical trip to India is a very simple process with India Cardiac Surgery Site
1. Fill in our enquiry form and one of our executives will contact you soon.
2. Call us at +91-9370586696 for any assistance.
3. Complete information regarding surgery is available on our website.
Echocardiography
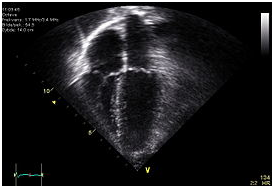
An echocardiogram. Image shows that the human heart has four chambers. Apical view – left side of the heart to the right. Anatomically correct image – heart’s apex at bottom. The trace in the lower left shows the cardiac cycle and the red mark the time in the cardiac cycle that the image was captured.
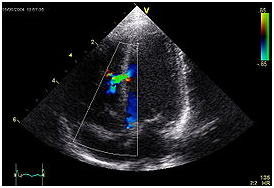
An abnormal echocardiogram. Image shows a mid-muscular ventricular septal defect. The trace in the lower left shows the cardiac cycle and the red mark the time in the cardiac cycle that the image was captured. Colors are used to represent the velocity and direction of blood flow.
An echocardiogram is a sonography of the heart. Also known as a cardiac ultrasound, it uses standard ultrasound techniques to image two-dimensional slices of the heart. The latest ultrasound systems now employ 3D real-time imaging.
In addition to creating two-dimensional pictures of the cardiovascular system, an echocardiogram can also produce accurate assessment of the velocity of blood and cardiac tissue at any arbitrary point using pulsed or continuous wave doppler ultrasound. This allows assessment of cardiac valve areas and function, any abnormal communications between the left and right side of the heart, any leaking of blood through the valves (valvular regurgitation), and calculation of the cardiac output as well as the Ejection fraction.
Echocardiography was an early medical application of ultrasound. Echocardiography was also the first application of intravenous contrast-enhanced ultrasound. This technique injects gas-filled microbubbles into the venous system to improve tissue and blood delineation. Contrast is also currently being evaluated for its effectiveness in evaluating myocardial perfusion. It can also be used with Doppler ultrasound to improve flow-related measurements (see Doppler echocardiography).
Echocardiography is usually performed by cardiac sonographers and interpreted by a cardiologist.
Purpose
Echocardiography is used to diagnose cardiovascular diseases. In fact, it is one of the most widely used diagnostic tests for heart disease. It can provide a wealth of helpful information, including the size and shape of the heart, its pumping capacity and the location and extent of any damage to its tissues. It is especially useful for assessing diseases of the heart valves. It not only allows doctors to evaluate the heart valves, but it can detect abnormalities in the pattern of blood flow, such as the backward flow of blood through partly closed heart valves, known as regurgitation. By assessing the motion of the heart wall, echocardiography can help detect the presence and assess the severity of coronary artery disease, as well as help determine whether any chest pain is related to heart disease. Echocardiography can also help detect hypertrophic cardiomyopathy, in which the walls of the heart thicken in an attempt to compensate for heart muscle weakness. The biggest advantage to echocardiography is that it is noninvasive (doesn’t involve breaking the skin or entering body cavities) and has no known risks or side effects.
Check out the Patient Testimonial, where the patient shares about their success stories from treatments through India Cardiac Surgery Consultants.
Transthoracic Echocardiogram
A standard echocardiogram is also known as a transthoracic echocardiogram (TTE), or cardiac ultrasound. In this case, the echocardiography transducer (or probe) is placed on the chest wall (or thorax) of the subject, and images are taken through the chest wall. This is a non-invasive, highly accurate and quick assessment of the overall health of the heart. A cardiologist can quickly assess a patient’s heart valves and degree of heart muscle contraction (an indicator of the ejection fraction). The images are displayed on a monitor, and are recorded either by videotape (analog) or by digital techniques.
An echocardiogram can be used to evaluate all four chambers of the heart. It can determine strength of the heart, the condition of the heart valves, the lining of the heart (the pericardium), and the aorta. It can be used to detect a heart attack, enlargement or hypertrophy of the heart, infiltration of the heart with an abnormal substance. Weakness of the heart, cardiac tumors, and a variety of other findings can be diagnosed with an echocardiogram. With advanced measurements of the movement of the tissue with time (tissue doppler), it can measure diastolic function, fluid status, and dys-synchrony.
The TTE is highly accurate for identifying vegetations (masses consisting of a mixture of bacteria and blood clots), but the accuracy can be reduced in up to 20% of adults because of obesity, chronic obstructive pulmonary disease, chest-wall deformities, or otherwise technically difficult patients. TTE in adults is also of limited use for the structures at the back of the heart, such as the left atrial appendage. Transesophageal echocardiography, if available, may be more accurate than TTE because it excludes the variables previously mentioned and allows closer visualization of common sites for vegetations and other abnormalities. Transesophageal echocardiography also affords better visualization of prosthetic heart valves.
Transesophageal Echocardiogram
This is an alternative way to perform an echocardiogram. A specialized probe containing an ultrasound transducer at its tip is passed into the patient’s esophagus. This allows image and Doppler evaluation which can be recorded. This is known as a transesophageal echocardiogram, or TEE (TOE in the United Kingdom). The advantage of TEE over TTE is usually clearer images, especially of structures that are difficult to view transthoracicly (through the chest wall). The explanation for this is the heart rests directly upon the esophagus leaving only millimeters in distance that the ultrasound beam has to travel. This reduces the attenuation (weakening) of the ultrasound signal, generating a stronger return signal, ultimately enhancing image and Doppler quality. Comparatively, transthoracic ultrasound must first traverse skin, fat, ribs and lungs before reflecting off the heart and back to the probe before an image can be created. All these structures, and the distance the beam must travel, attenuate the ultrasound signal, degrading image and Doppler quality.
In adults, several structures can be evaluated and imaged better with the TEE, including the aorta, pulmonary artery, valves of the heart, both atria, atrial septum, left atrial appendage, and coronary arteries. While TTE can be performed quickly, easily and without pain to the patient, TEE requires a fasting patient, a team of medical personnel, takes longer to perform, is uncomfortable for the patient and has some risks associated with the procedure (esophageal perforation–1 in 10,000, and adverse reactions to the medication).
Before inserting the probe, conscious sedation is induced with the patient to ease the discomfort of the individual and to decrease the gag reflex, thus making the ultrasound probe easier to pass into the esophagus. Conscious sedation is a light sedation usually using the medications midazolam (a benzodiazepine with sedating, amnesiac qualities) and fentanyl. Sometimes a local anesthetic spray is used for the back of the throat, such a xylocaine and/or a jelly/lubricant anesthetic for the esophagus. Children are anesthetized. Unlike the TTE, the TEE is considered an invasive procedure and is thus performed by physicians in the U.S., not sonographers.
3-Dimensional Echocardiography
3D echocardiogram of a heart viewed from the apex
3-D echocardiography is now possible, using an ultrasound probe with an array of transducers and an appropriate processing system. This enables detailed anatomical assessment of cardiac pathology, particularly valvular defects, cardiomyopathies, and congenital abnormalities.
Aortic Valve Area Calculation
Aortic valve area calculation is an indirect method of determining the area of the aortic valve. The calculated aortic valve orifice area is currently one of the measures for evaluating the severity of aortic stenosis. A valve area of less than 0.8 cm² is considered to be severe aortic stenosis.
There are many ways to calculate the valve area of aortic stenosis. The most commonly used methods involve measurements taken during echocardiography. For interpretation of these values, the area is generally divided by the body surface area, to arrive at the patient’s optimal aortic valve orifice area.
Planimetry
Planimetry is the tracing out of the opening of the aortic valve in a still image obtained during echocardiographic acquisition during ventricular systole, when the valve is supposed to be open. While this method directly measures the valve area, the image may be difficult to obtain due to artifacts during echocardiography, and the measurements are dependent on the technician who has to manually trace the perimeter of the open aortic valve. Because of these reasons, planimetry of aortic valve is not routinely performed.
The Continuity Equation
The continuity equation states that the flow in one area must equal the flow in a second area if there are no shunts in between the two areas. In practical terms, the flow from the left ventricular outflow tract (LVOT) is compared to the flow at the level of the aortic valve. Using Echocardiography, the aortic valve area calculated using the time velocity integral (TVI) is most accurate method and is the preferred method. The flow through the LVOT, or LV Stroke Volume (cm3 or cc), can be calculated by measuring the LVOT diameter (cm), squaring that value, multiplying the value by 0.78540 giving cross sectional area of the LVOT (cm2)and multiplying that value by the LVOT TVI (cm), measured on the spectral Doppler display using pulsed-wave Doppler. From these, it is easy to calculate the area (cm2) of the aortic valve by simply dividing the LV Stroke Volume (cm3) by the AV TVI (cm) measured on the spectral Doppler display using continuous-wave Doppler.
Example: An individual undergoes transthoracic echocardiography for the evaluation of a systolic ejection murmur with delayed carotid upstroke noted on physical examination. During echocardiography, the following measurements were made: LVOT diameter of 2 cm, LVOT TVI of 24 cm, and an Aortic Valve TVI of 50 cm. What is the aortic valve area?
Answer: An LVOT diameter of 2 cm gives a LVOT cross-sectional area of, 2 * 2 * 0.78540 = 3.14 cm2. To calculate stroke volume, multiply the cross-sectional area of 3.14 cm2 by the LVOT TVI 24 cm. This gives an LV Stroke Volume of 3.14 * 24 = 75.40 cc. Divide the LV Stroke Volume, 75.40 cc by the Aortic Valve TVI, 50 cm and this gives an aortic valve area of 75.40 / 50 = 1.51 cm2.
The weakest aspect of this calculation is the variability in measurement of LVOT area, because it involves squaring the LVOT dimension. Therefore, it is crucial for the sonographer to take great care in measuring the LVOT diameter.
The Gorlin Equation
The Gorlin equation states that the aortic valve area is equal to the flow through the aortic valve during ventricular systole divided by the systolic pressure gradient across the valve times a constant. The flow across the aortic valve is calculated by taking the cardiac output (measured in liters/minute) and dividing it by the heart rate (to give output per cardiac cycle) and then dividing it by the systolic ejection period measured in seconds per beat (to give flow per ventricular contraction).
The Gorlin equation is related to flow across the valve. Because of this, the valve area may be erroneously calculated as stenotic if the flow across the valve is low (ie: if the cardiac output is low). The measurement of the true gradient is accomplished by temporarily increasing the cardiac output by the infusion of positive inotropic agents, such as dobutamine.
Example: An individual undergoes left and right heart cardiac catheterization as part of the evaluation of aortic stenosis. The following hemodynamic parameters were measured. With a heart rate of 80 beats/minute and a systolic ejection period of 0.33 seconds, the cardiac output was 5 liters/minute. During simultaneous measurement of pressures in the left ventricle and aorta (with the use of one catheter in the left ventricle and a second in the ascending aorta), the mean systolic pressure gradient was measured at 50 mmHg. What is the valve area as measured by the Gorlin equation?
The Hakki Equation
The Hakki equation is a simplification of the Gorlin equation, relying on the observation that in most cases, the numerical value of
The resulting simplified formula is:
Example: An individual undergoes left and right cardiac catheterization for the evaluation of aortic stenosis. Measurements includes an aortic pressure of 120/60, LV pressure of 170/15, cardiac output of 3.5 liters/minute. What is the aortic valve area?
Answer: The peak gradient between the LV and aorta is 50 mmHg. This gives
Accreditation
The “Intersocietal Commission for the Accreditation of Echocardiography Laboratories” (ICAEL) sets standards for labs and technicians in the US to comply to. Once all requirements have been met, the lab or technician will receive ICAEL certification. A lab that has received the certification may also receive higher reimbursement from insurance companies such as Medicare.
In the UK, accreditation is done by the British Society of Echocardiography. Accredited technicians or other professionals from the echocardiography field will have completed a logbook and passed an exam. A useful website to learn more about real time 3 dimensional echocardiography and an international portal for uploading images for discussion is www.3dechocardiography.com
Aortic Valve Replacement
Aortic valve replacement is a cardiac surgery procedure in which a patient’s aortic valve is replaced by a different valve. The aortic valve can be affected by a range of diseases; the valve can either become leaky (aortic insufficiency / regurgitation) or partially blocked (aortic stenosis). Aortic valve replacement currently requires open heart surgery.
Types of Heart Valves
There are two basic types of artificial heart valve: mechanical valves and tissue valves.
- Tissue valves
Tissue heart valves are usually made from animal tissues, either animal heart valve tissue or animal pericardial tissue. The tissue is treated to prevent rejection and calcification.There are alternatives to animal tissue valves. In some cases a homograft – a human aortic valve — can be implanted. Homograft valves are donated by patients and harvested after the patient dies. The durability of homograft valves is probably the same for porcine tissue valves. Another procedure for aortic valve replacement is the Ross procedure (or pulmonary autograft). In a Ross procedure, the aortic valve is removed and replaced with the patient’s own pulmonary valve. A pulmonary homograft (pulmonary valve taken from a cadaver)is then used to replace the patient’s own pulmonary valve. This procedure was first used in 1967 and is used primarily in children.
- Mechanical valvesMechanical valves are designed to outlast the patient, and have typically been stress-tested to last several hundred years. Although mechanical valves are long-lasting and generally only one surgery is needed, there is an increased risk of blood clots forming with mechanical valves. As a result, mechanical valve recipients must generally take anti-coagulant (blood thinning) drugs such as warfarin for the rest of their lives, which makes the patient more prone to bleeding.
Valve selection
Tissue valves tend to wear out faster with increased flow demands – such as with a more active (typically younger person). Tissue valves typically last 10-15 years in less active (typically elderly) patients, but wear out faster in younger patients. When a tissue valve wears out and needs replacement, the person must undergo another valve replacement surgery. For this reason, younger patients are often recommended mechanical valves to prevent the increased risk (and inconvenience) of another valve replacement.
Surgical Procedure
Aortic valve replacement is most frequently done through a median sternotomy, meaning the chestbone is sawed in half. Once the pericardium has been opened, the patient is placed on cardiopulmonary bypass machine, also referred to as the heart-lung machine. This machine takes over the task of breathing for the patient and pumping his blood around while the surgeon replaces the heart valve.
Once the patient is on bypass, an incision is made in the aorta. The surgeon then removes the patient’s diseased aortic valve and a mechanical or tissue valve is put in its place. Once the valve is in place and the aorta has been closed, the patient is taken off the heart-lung machine. Transesophageal echocardiogram (TEE, an ultra-sound of the heart done through the esophagus) can be used to verify that the new valve is functioning properly. Pacing wires are usually put in place, so that the heart can be manually paced should any complications arise after surgery. Drainage tubes are also inserted to drain fluids from the chest and pericardium following surgery. These are usually removed within 36 hours while the pacing wires are generally left in place until right before the patient is discharged from the hospital.
Hospital Stay and Recovery Time
Immediately after aortic valve replacement, the patient will frequently stay in a cardiac surgery intensive care unit for 12-36 hours. After this, the patient is often moved to a lower-dependency unit and then to a cardiac surgery ward. Total time spent in hospital following surgery is usually between 4 and 10 days, unless complications arise.
Recovery from aortic valve replacement will take 1-3 months if the patient is in good health. Patients are advised not to do any heavy lifting for 6-8 weeks following surgery to avoid damaging the sternum (breast bone) while it heals.
Surgical Outcome and Risk of Procedure
The risk of death or serious complications from aortic valve replacement are typically quoted as being between 1-5%, depending on the health and age of the patient, as well as the skill of the surgeon. Older patients, as well as more fragile ones, are sometimes ineligible for surgery because of elevated risks.
Future Developments
Percutaneous aortic valve replacement, which allows the implantation of valves using a catheter without open heart surgery is still being evaluated in clinical trials and appears to be promising in patients who are at high risk to undergo open heart surgery.
If you are really seeking for Echocardiogram in India, kindly fill up the form for a free consultation with our expert cardiologists. You will be provided with thorough analysis and suggestions regarding the Echocardiogram you are seeking for.
Tags
cardiac procedures, affordable echocardiogram procedure in india, echocardiogram in india, echocardiogram procedure in india, echocardiogram procedure in india reviews, free consultation echocardiogram procedure in india, low cost echocardiogram procedure in india

